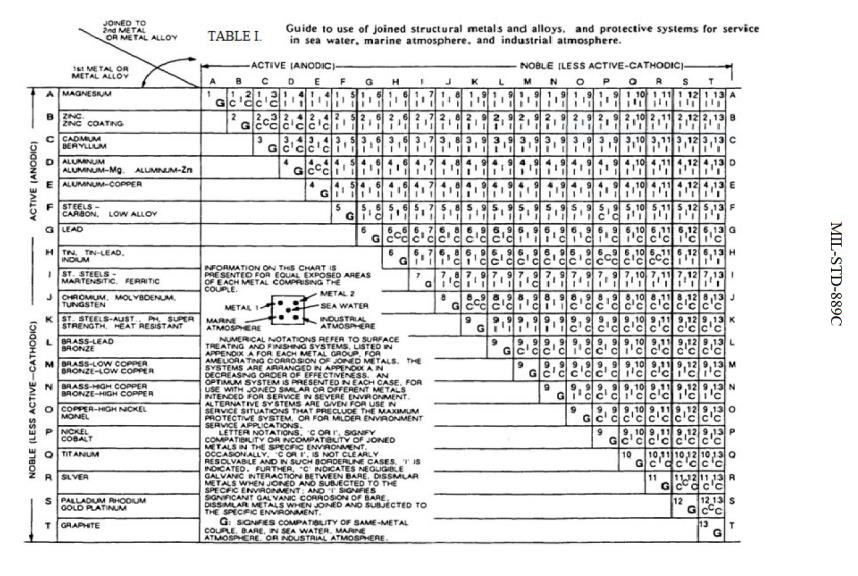
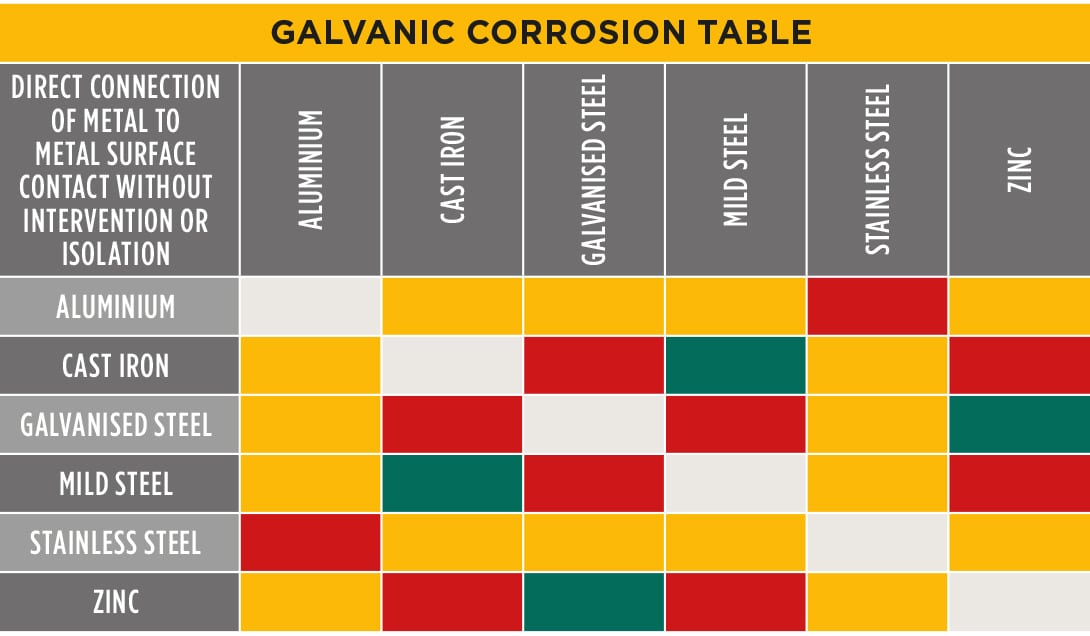
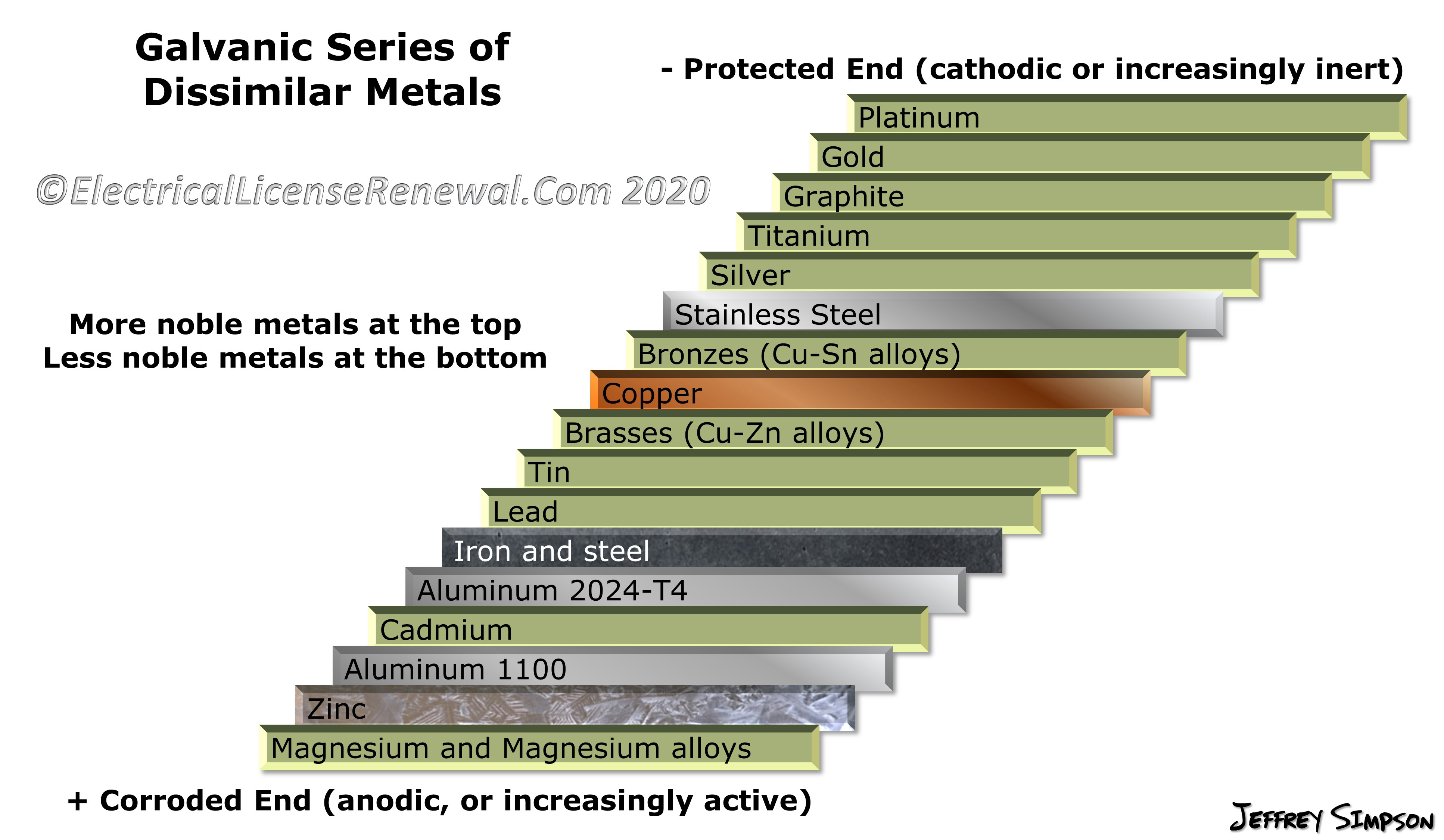
Dissimilar Metal Corrosion Chart
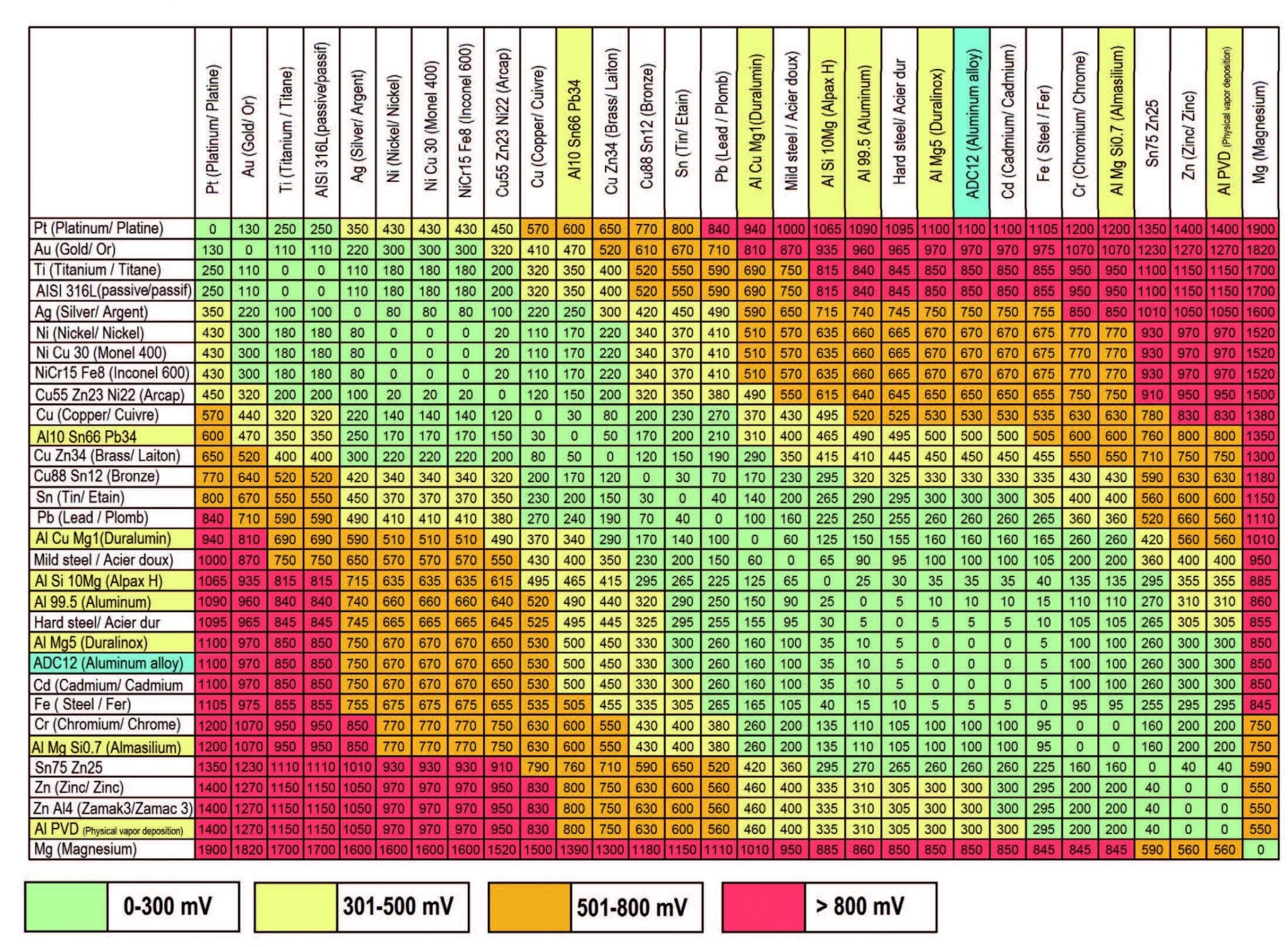
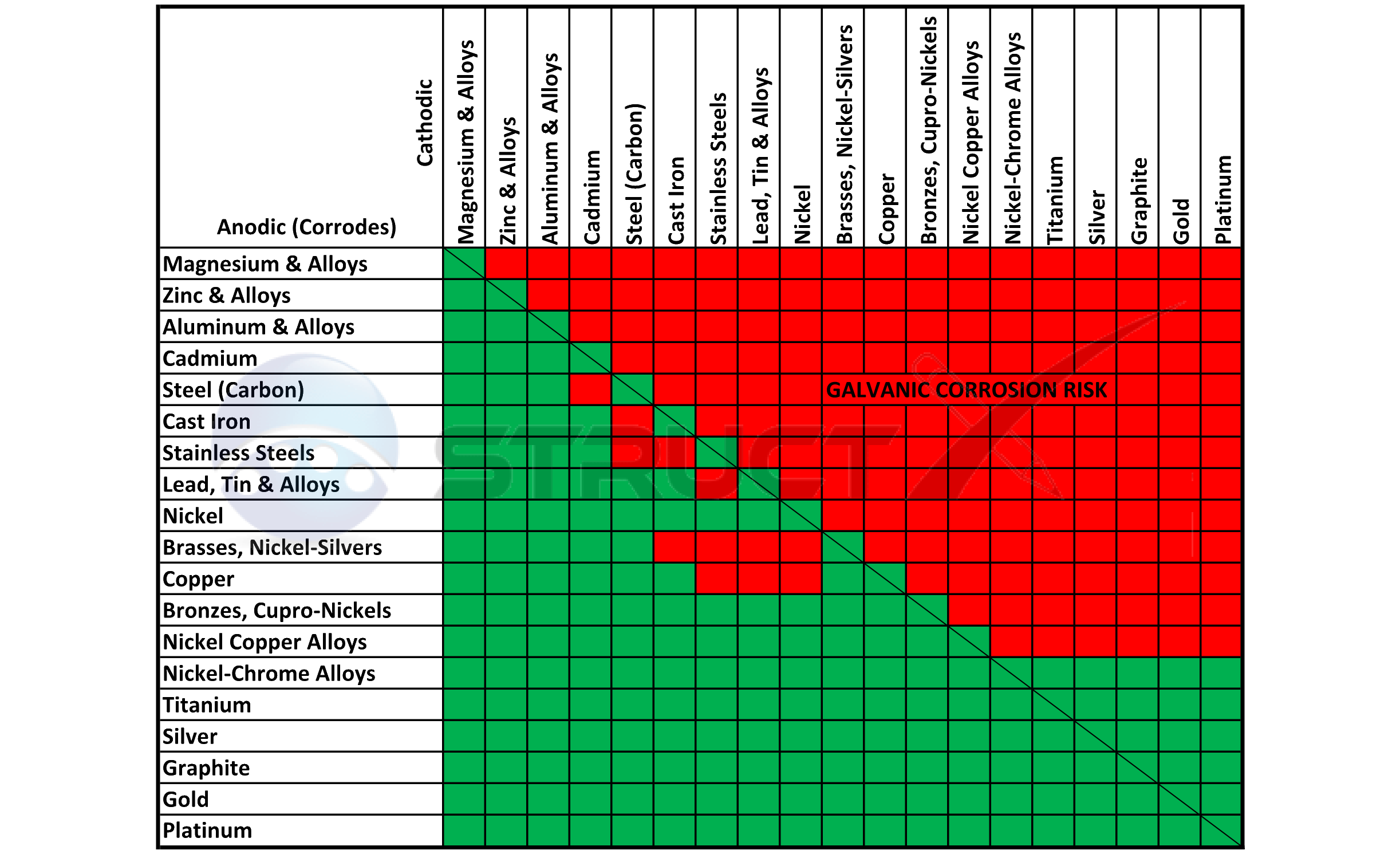
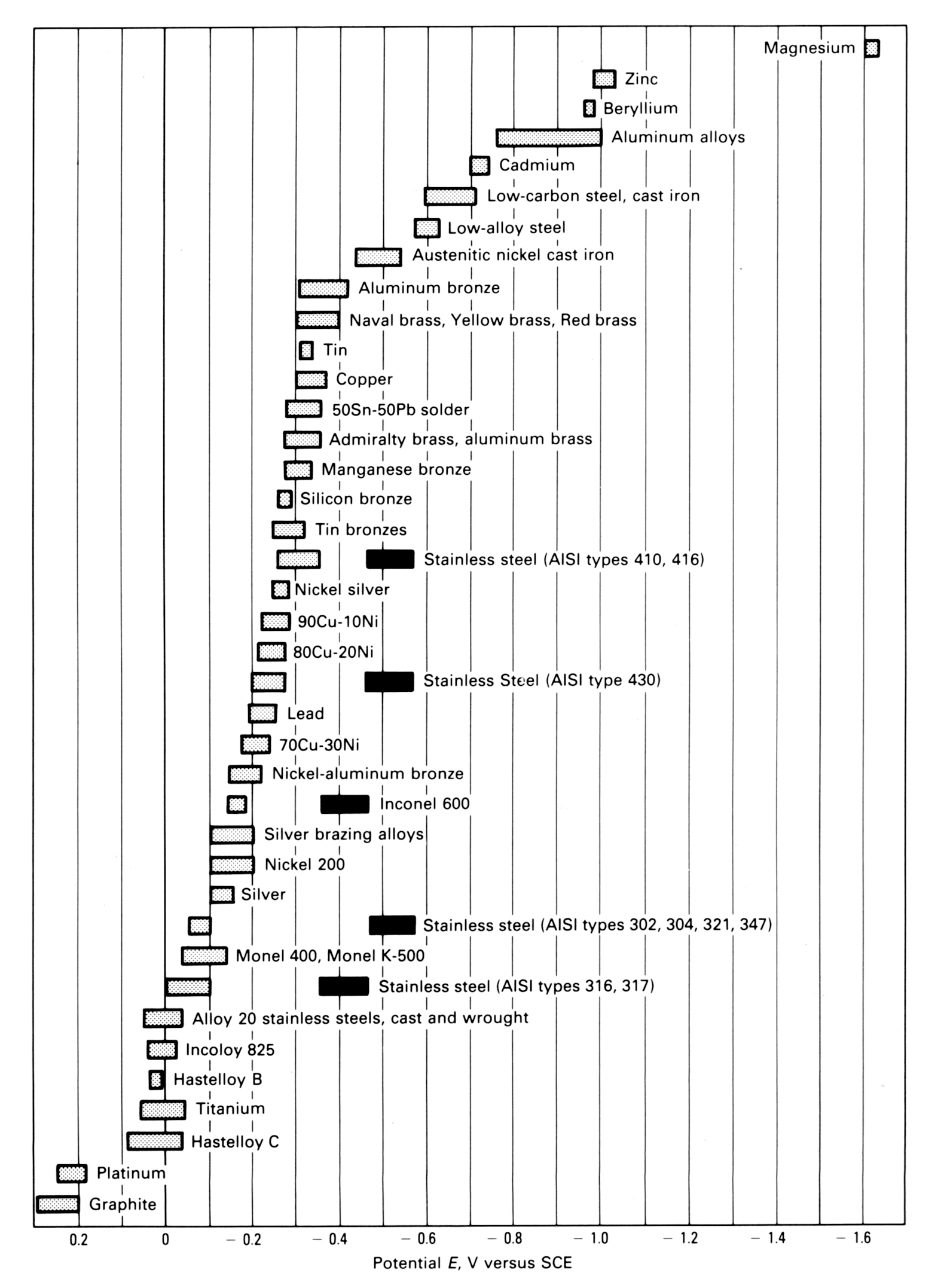
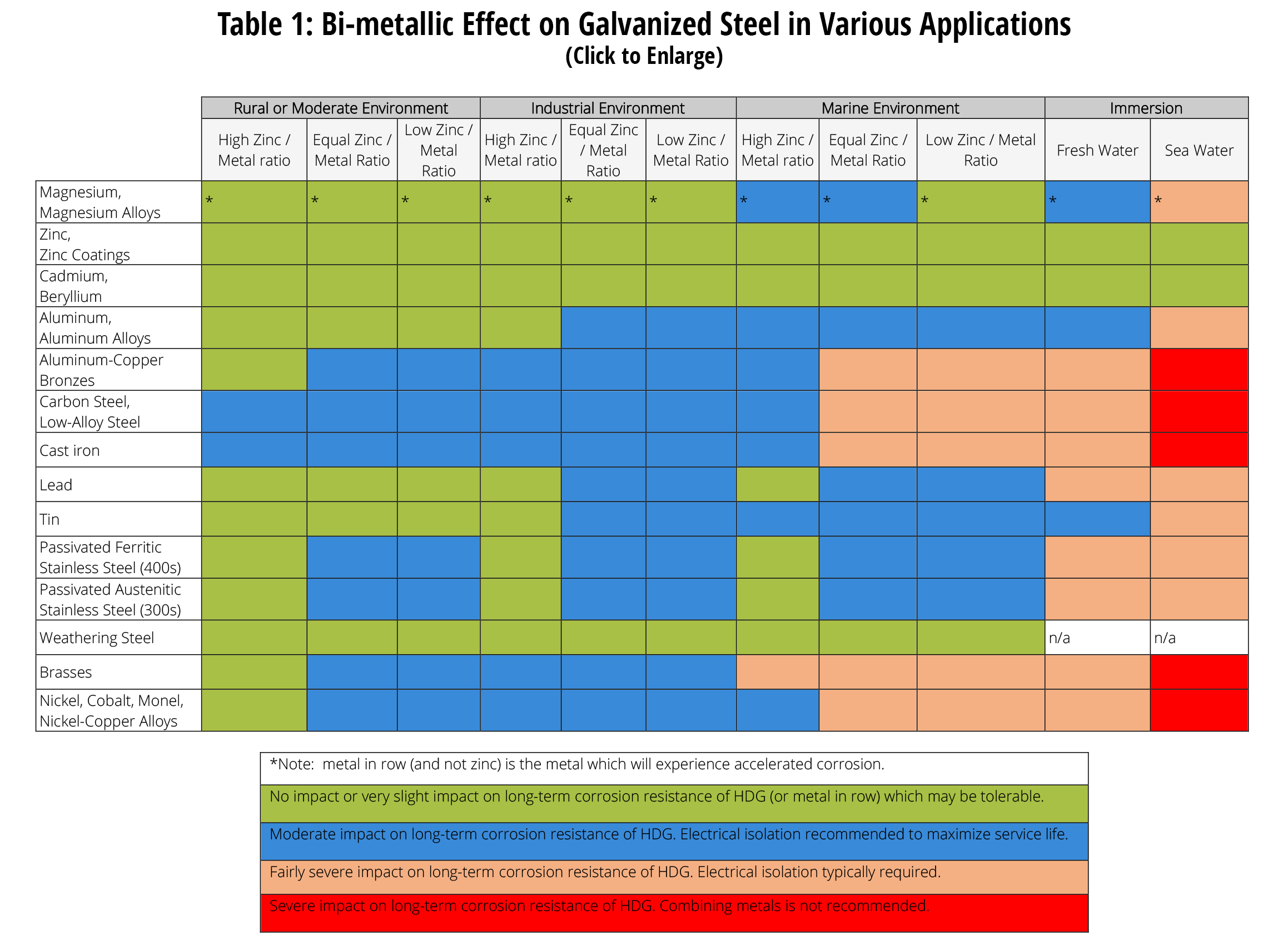
Dissimilar Metal Corrosion Chart - Web this article examines how dissimilar metals can lead to galvanic corrosion. It may also take place with one metal with. Use this chart to avoid galvanic corrosion in seawater when different metals come in to contact. Web below is a galvanic reaction chart for dissimilar metals. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. Web below, we delve into dissimilar metal corrosion, how and why it occurs, and tips for avoiding it to prevent accidents and damaging project delays. Web learn how to prevent galvanic corrosion (dissimilar metal corrosion) by understanding the three conditions that must exist for it to occur. So, for example, choosing zinc on zinc would have the lowest risk for. It includes a chart that shows how different plating materials react to one another with. This phenomenon is named after italian ph… A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. This phenomenon is named after italian ph… So, for example, choosing zinc on zinc would have the lowest risk for. Web galvanic corrosion typically attacks junction areas of dissimilar metals and occurs when the following three conditions are met. Web below is a galvanic reaction chart for dissimilar metals. Web find out how dissimilar metals will corrode when placed against each other in an assembly using this chart. Web below, we delve into dissimilar metal corrosion, how and why it occurs, and tips for avoiding it to prevent accidents and damaging project delays. Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal. Web galvanic corrosion occurs when two dissimilar metals are in contact electrically in the presence of an electrolyte. Web however, you can completely avoid galvanic corrosion by choosing matching metal anchors. Web galvanic corrosion occurs when two dissimilar metals are in contact electrically in the presence of an electrolyte. For any combination of dissimilar metals, the metal with the lower number will act as an anode and will corrode. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. Web the. Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. Web learn how to prevent galvanic corrosion (dissimilar metal corrosion) by understanding the three conditions that must exist for it to occur. Web however, you can. Web below, we delve into dissimilar metal corrosion, how and why it occurs, and tips for avoiding it to prevent accidents and damaging project delays. Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal. So, for example, choosing zinc on zinc would have. Web however, you can completely avoid galvanic corrosion by choosing matching metal anchors. This phenomenon is named after italian ph… It may also take place with one metal with. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. Web electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices. Web find out how dissimilar metals will corrode when placed against each other in an assembly using this chart. Web below is a galvanic reaction chart for dissimilar metals. This chart is designed to assist in broadly assessing the risk of. Web find out how dissimilar metals will corrode when placed against each other in an assembly using this chart. It includes a chart that shows how different plating materials react to one another with. Web galvanic corrosion typically attacks junction areas of dissimilar metals and occurs when the following three conditions are met. Web galvanic corrosion occurs when two dissimilar. Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. Find a table of anodic index values for. Web below, we delve into dissimilar metal corrosion, how and why it occurs, and tips for avoiding it. Web this article examines how dissimilar metals can lead to galvanic corrosion. Web galvanic corrosion (also called ' dissimilar metal corrosion' or wrongly 'electrolysis') refers to corrosion damage induced when two dissimilar materials are coupled in a corrosive. Web electrolytic corrosion (electrolysis) occurs when dissimilar metals are in contact in the presence of an electrolyte, such as water (moisture) containing. Web the susceptibility of different base metals to corrosion while in contact depends upon the difference between the contact potentials or the electromotive voltages of the metals. This phenomenon is named after italian ph… If two different metals are placed in electrical contact and bridged by an electrolyte, a current figure 1: It may also take place with one metal. It includes a chart that shows how different plating materials react to one another with. Web this article examines how dissimilar metals can lead to galvanic corrosion. Web when design requires that dissimilar metals come in contact, galvanic compatibility can be managed by finishes and plating which protects the base materials from corrosion. The chart shows the galvanic corrosion potential. This phenomenon is named after italian ph… It may also take place with one metal with. Web galvanic corrosion (also called ' dissimilar metal corrosion' or wrongly 'electrolysis') refers to corrosion damage induced when two dissimilar materials are coupled in a corrosive. Galvanic corrosion (also called bimetallic corrosion or dissimilar metal corrosion) is an electrochemical process in which one metal corrodes preferentially when it is in electrical contact with another, in the presence of an electrolyte. The chart shows the galvanic corrosion potential of various metals. During this process, corrosion occurs on the anode, whereas. Web the susceptibility of different base metals to corrosion while in contact depends upon the difference between the contact potentials or the electromotive voltages of the metals. This chart is designed to assist in broadly assessing the risk of galvanic corrosion associated with a given metal coming. Web below, we give a brief overview of galvanic corrosion and provide a galvanic corrosion chart to help fabricators and machinists avoid using the wrong metal. Use this chart to avoid galvanic corrosion in seawater when different metals come in to contact. So, for example, choosing zinc on zinc would have the lowest risk for. For any combination of dissimilar metals, the metal with the lower number will act as an anode and will corrode. Web when design requires that dissimilar metals come in contact, galvanic compatibility can be managed by finishes and plating which protects the base materials from corrosion. If two different metals are placed in electrical contact and bridged by an electrolyte, a current figure 1: Web galvanic corrosion typically attacks junction areas of dissimilar metals and occurs when the following three conditions are met. A similar galvanic reaction is exploited in primary cells to generate a useful electrical voltage to power portable devices.Galvanic Corrosion Chart Dissimilar Metals A Visual Reference of
Dissimilar Metal Corrosion Chart
Galvanic Corrosion Chart Dissimilar Metals
Dissimilar joining of Al with steel? r/Welding
FAQ 1 Galvanic/Dissimilar Metal Corrosion
Dissimilar Metal Corrosion with… American Galvanizers Association
Dissimilar metal corrosion with chemical filmed (Alodine, Iridite
Dissimilar Metals Corrosion Chart
Dissimilar Corrosion Materials Tables
Stainless Steel Galvanic Corrosion Chart
Web Electrolytic Corrosion (Electrolysis) Occurs When Dissimilar Metals Are In Contact In The Presence Of An Electrolyte, Such As Water (Moisture) Containing Very Small Amounts Of.
Web Find Out How Dissimilar Metals Will Corrode When Placed Against Each Other In An Assembly Using This Chart.
Web Galvanic Series, Or Nobility Chart For Dissimilar Metals.
Web Galvanic Corrosion Occurs When Two Dissimilar Metals With Different Potentials Are Placed In Electrical Contact In An Electrolyte.
Related Post: